The fundamental challenge in Alzheimer's disease (AD) prognostication lies not in population-level predictions but in individual-level trajectory forecasting. Clinical progression varies dramatically across patients—some experience rapid cognitive deterioration while others plateau for years—making generic prognostic models clinically inadequate. Traditional regression approaches fail to capture this heterogeneity, and existing machine learning solutions often exhibit demographic bias or collapse entirely when confronted with the realistic messiness of clinical data. This heterogeneity problem becomes acute when designing clinical trials, where enrichment strategies require precise identification of patients likely to show measurable decline within trial windows. The CognitiveTwin framework addresses these interconnected challenges through a principled probabilistic architecture designed explicitly for longitudinal, multimodal clinical data.
The clinical stakes are substantial. Reliable personalized predictions enable more efficient trial designs, reduce sample sizes through better patient stratification, and support shared decision-making in clinical settings. Yet achieving this reliability demands solving three distinct technical problems simultaneously: maintaining prediction accuracy across diverse patient populations, ensuring demographic fairness (preventing systematic bias against underrepresented groups), and gracefully degrading rather than catastrophically failing when data is missing-not-at-random (MNAR)—a ubiquitous problem in real clinical cohorts where dropout correlates with disease severity.
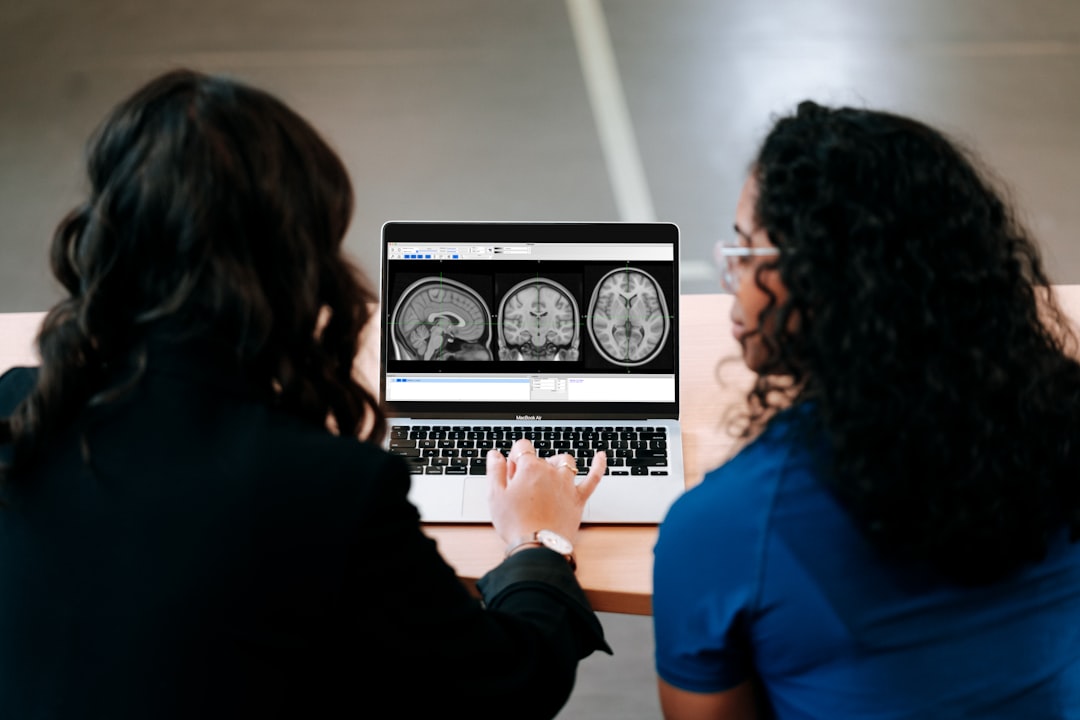
The CognitiveTwin architecture integrates five distinct data modalities: cognitive assessment scores (Mini-Cog, MMSE variants), structural and functional magnetic resonance imaging (MRI), amyloid and tau positron emission tomography (PET), cerebrospinal fluid (CSF) biomarkers (phosphorylated tau, amyloid-β42), and genetic risk factors (APOE ε4 status and polygenic risk scores). The fusion mechanism employs a Transformer-based encoder that processes each modality independently before learning cross-modal attention weights, enabling the model to dynamically weight modality importance for individual patients. This design choice is critical: different patients exhibit different biomarker profiles (some are PET-positive but cognitively normal; others show cognitive decline without amyloid pathology), so learned attention mechanisms can capture these heterogeneous patterns better than fixed fusion weights.
The temporal dynamics component leverages Deep Markov Models (DMMs), which factorize the joint distribution of observations and latent states through a hierarchical structure: p(x₁:T, z₁:T) = p(z₁)∏ₜp(zₜ|zₜ₋₁)p(xₜ|zₜ). This formulation allows the model to learn interpretable latent disease states while generating patient-specific trajectories. Unlike RNNs that maintain deterministic hidden states, DMMs explicitly model uncertainty over state transitions, crucial for capturing the stochastic nature of neurodegeneration. The model was trained on 1,666 patients from the TADPOLE challenge dataset (derived from the Alzheimer's Disease Neuroimaging Initiative), with longitudinal follow-up spanning 5-10 years.
The evaluation protocol addresses the three critical dimensions. Prediction accuracy was assessed via standard metrics (RMSE, correlation coefficients) on held-out test sets. Fairness was quantified by measuring demographic parity and equalized odds across age, sex, and racial/ethnic groups—examining whether prediction error distributions diverge significantly across subpopulations. Critically, robustness to MNAR missingness was evaluated by synthetically introducing missing data patterns that correlate with outcomes (simulating clinical dropout where sicker patients miss appointments), then measuring whether performance degrades gracefully.
The framework's demonstrated resilience to MNAR patterns is particularly noteworthy. Standard imputation methods (mean-filling, K-nearest neighbors) introduce bias when missingness is informative, but CognitiveTwin's probabilistic formulation allows it to marginalize over missing values rather than impute them, preserving uncertainty quantification. This is essential for clinical deployment, where acknowledging prediction uncertainty is ethically mandatory.
CuraFeed Take: CognitiveTwin represents a maturation of digital twin methodology for neurodegenerative disease—moving beyond proof-of-concept toward clinically deployable systems. The explicit treatment of fairness and MNAR robustness signals that the authors understand deployment realities; many academic papers achieve accuracy on clean test sets then fail catastrophically in clinical settings. However, several questions warrant scrutiny: First, how does performance generalize to datasets outside TADPOLE? The cohort skews toward higher socioeconomic status and cognitive reserve, potentially limiting applicability to diverse real-world populations. Second, the Transformer-DMM architecture adds significant complexity—ablation studies comparing against simpler baselines (linear mixed models, standard LSTMs) would clarify whether this complexity yields commensurate gains or represents over-engineering. Third, the clinical utility hinges on whether predictions improve trial enrichment beyond simpler biomarker cutoffs; this requires prospective validation, not retrospective analysis.
The broader implication is that precision medicine in neurodegenerative disease increasingly demands probabilistic, multimodal architectures that respect data missingness and demographic heterogeneity. This work may catalyze similar approaches in Parkinson's disease and frontotemporal dementia, where similar heterogeneity challenges exist. Watch for follow-up work on: (1) prospective validation in independent cohorts, (2) interpretability analysis of learned latent states and attention patterns, and (3) integration with real-time wearable data to extend beyond clinic-based biomarkers. The fairness-by-design approach here should become table stakes for clinical AI, not a differentiator.